What Does a Catalytic Converter Do?
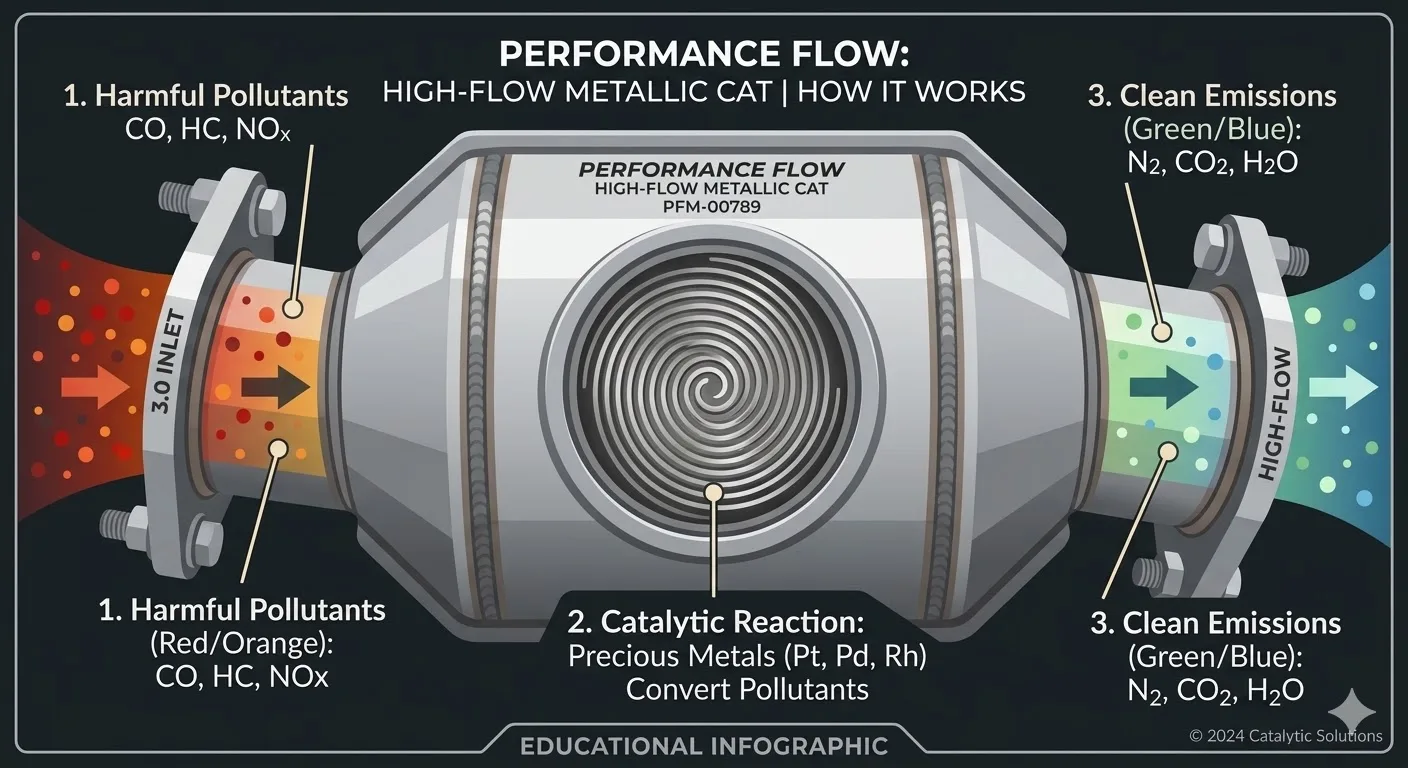
A catalytic converter is a device in your car's exhaust system that converts toxic pollutants into less harmful gases before they exit the tailpipe. Think of it as an environmental filter for your car's exhaust — it takes the dangerous byproducts of burning gasoline (carbon monoxide, hydrocarbons, and nitrogen oxides) and transforms them into safer substances (carbon dioxide, water vapor, and nitrogen).
The converter achieves this through catalytic chemical reactions — meaning the precious metals inside (platinum, palladium, rhodium) trigger chemical changes in the exhaust gases without being consumed themselves. The word "catalyst" literally means something that causes a reaction without being changed by it.
How It Works in 4 Steps
Step 1: Your engine burns gasoline and air, producing exhaust gases that include toxic compounds. Step 2: These hot gases flow through the exhaust manifold and into the catalytic converter. Step 3: Inside the converter, gases pass through a honeycomb structure coated with precious metals. The metals trigger chemical reactions that break apart harmful molecules and recombine them into safer ones. Step 4: The cleaned exhaust exits through the muffler and tailpipe.
Why Every Gas Car Has One
Federal law has required catalytic converters on all gasoline-powered vehicles sold in the US since 1975. Without converters, modern vehicles would release approximately 90% more harmful emissions, contributing significantly to smog, acid rain, and respiratory health problems. The catalytic converter is considered one of the most important environmental inventions in automotive history.
The Three Reactions
Modern three-way catalytic converters perform three simultaneous reactions: 1) Reduction of nitrogen oxides (NOx → N₂ + O₂), breaking apart smog-causing nitrogen oxide molecules. 2) Oxidation of carbon monoxide (CO → CO₂), converting the deadly odorless gas into carbon dioxide. 3) Oxidation of hydrocarbons (HC → CO₂ + H₂O), burning off unburned fuel particles into carbon dioxide and water.